Anodic Oxidation
The discussion on environmental and climate protection became one of the key drivers in the field of research and development. As a result, lightweight design is becoming increasingly important for saving energy and resources, especially in engineering and construction. In addition to plastics, which often fail in terms of strength and thermal stability, light metals such as titanium, aluminum and magnesium are enjoying growing popularity. In order to meet the high demands of the various application areas, e.g. aerospace, automotive or implant industries, these materials require both functional and decorative surfaces as well as resistant and corrosion-proof protection layers.
A special advantage of the light metals mentioned is their natural characteristic to get coated by a protective oxide layer passivating the surface. For the first time, H. Buff observed this feature for aluminum in 1857 and laid a decisive foundation to establish the industrial procedure.
The process of converting a metal surface into its metal oxides and hydroxides forced by application of an external current flow is called anodic oxidation. The component used as an anode is connected to a conductive, normally aqueous solution (electrolyte) and an inert, unchangeable cathode forming a closed electric circuit. The passive protective layers generated by this process can reach thicknesses of a few nanometers (natural oxide layer for aluminum is about 1.5–5 nm) up to several micrometers, e.g. hard anodized up to 250 µm, and convince with a very good adhesion.
ELOXAL – Anodizing
In 1927, the VAW (Vereinigte Aluminium-Werke AG) coined the term ELOXAL for anodizing aluminum and today, this process is still one of the most important methods for finishing aluminum surfaces. Depending on the reaction parameters selected it is possible to create layers with different morphologies and surface properties, ranging from barrier-like, compact to highly porous. Especially the porous layers can be additionally colored with organic and inorganic pigments in almost all shades for decorative purposes.
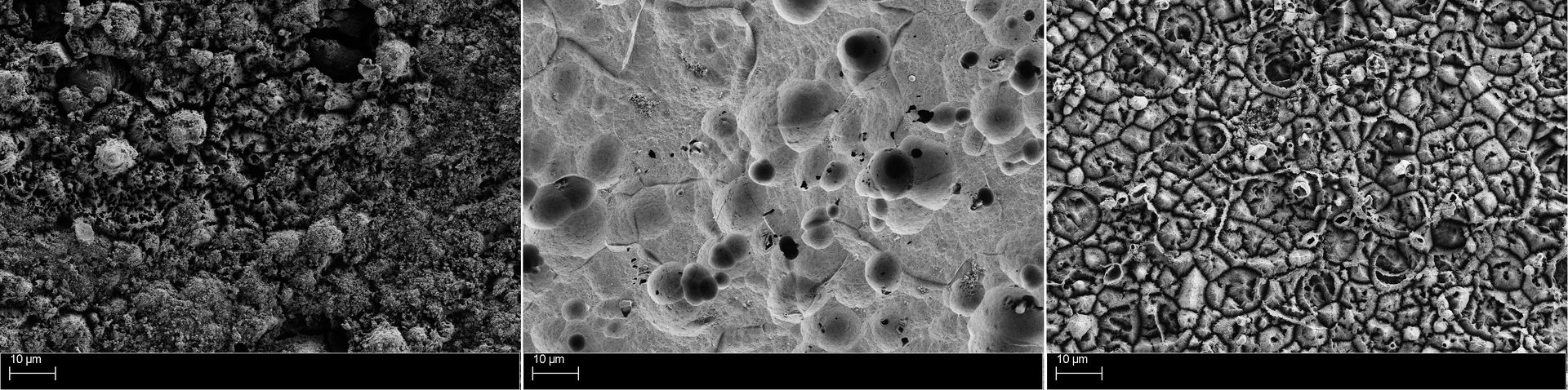
The increasing interest of the industry in the further development and optimization of the anodizing process caused INNOVENT the installation of an own laboratory equipment for anodizing aluminum surfaces. In addition to the common DC technology, which is used in the standard sulfuric acid direct current process (GS process), we are also able to anodize by alternating current and pulse technology, opening up a completely new field of research.
PCO® Process

The PCO® process is another special application of the anodic oxidation. This process is positioned at the interface of electrochemistry and plasma physics and is based on the formation of ceramic layers by spark discharge. These discharges produce a plasma-like state on the surface of the metal partial melting the top layer. This special process control allows the variation and adjustment of the surface properties within wide limits.
The PCO® process has been established at INNOVENT since 1999. Here you can find further information about the process and its abilities for your surfaces.




