Oleophobization
The wetting of solids with liquids – often characterized by the resulting contact angle – depends on surface and interfacial energies, special chemical interactions as well as topological conditions (such as roughness).
Due to their low surface energy, silicones oils and hydrocarbon oils wet most surfaces very well. At a correspondingly low viscosity, this wetting behavior is shown by "running / widening" (so-called spreading). This effect is desirable for corrosion protection or lubrication. However, if it concerns technological processes, which require "clean" surfaces, the possible contamination caused by the spreading of oil is undesirable and disturbing.
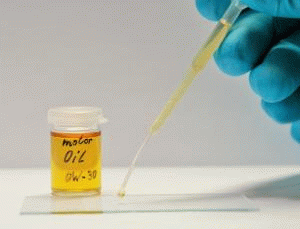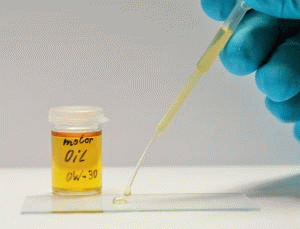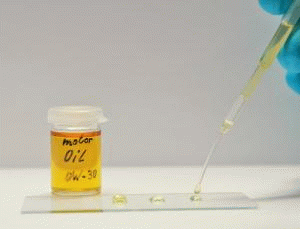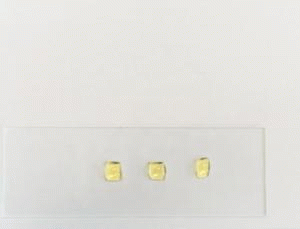
Fluorocarbon compounds with perfluorinated chains allow the creation of extremely low-interaction, i.e. largely non-polar and non-polarizable surfaces. As a result, these surfaces exhibit besides the oleophobic also highly hydrophobic properties. The wettability of such surfaces with hydrocarbon oils, silicone oils and many other substances is greatly reduced so that spreading can be effectively stopped.
In addition to their function as spread stoppers, surfaces oleophobized with fluorocarbon compounds show a decreased tendency to soiling and are easier to clean. This effect is used, e.g. to finish textiles or to provide surfaces with an anti-graffiti finish.
A current research area is the combination of very high hydrophilicity with oleophobic properties. With the appropriate setting, it can be achieved that adhering oils and fats can easily be undermined by water and be released / delaminate from the surface. Thus, the use of surfactants and tensides can be reduced or avoided.
We are happy to assist you in the selection and formulation of oleophobic coating materials for your application and are available for questions and comments.

Dr. Joerg Leuthaeusser
Head of Department
Primer and Chemical Surface Treatment
e-mail
Phone: +49 3641 2825 48